My healthcare practice is located in USA, why do I need a Consent Management app on my healthcare website?
A Consent Management App on your healthcare website is essential for ensuring compliance with various privacy and data protection regulations, especially HIPAA in the USA. It allows your practice to manage and record patients’ consent for data collection, communication, and the use of their protected health information (PHI). Even from the initial patient contact from your website, you still need to be able to obtain consent, and keep it for up to 6 years.
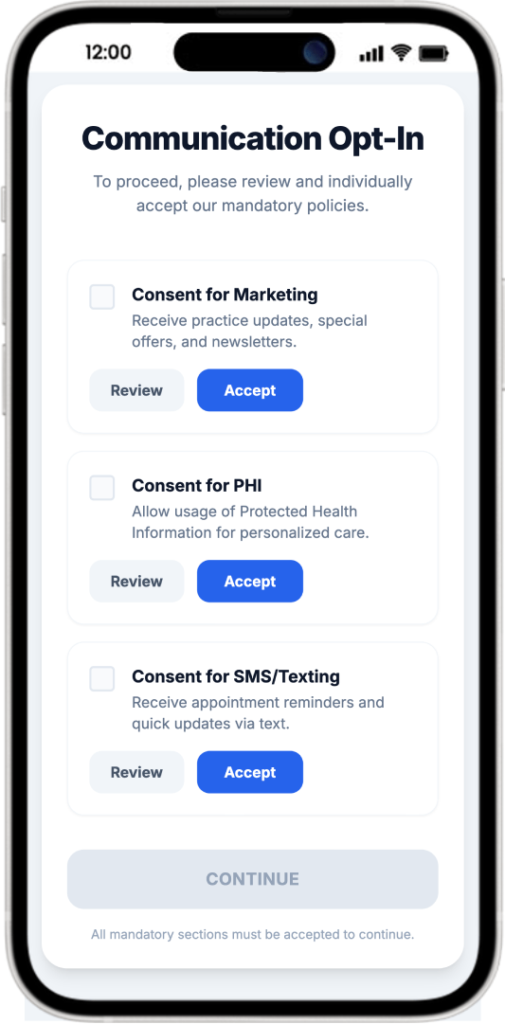
Under HIPAA, any identifiable health information—including data collected via website contact forms, SMS/Text messages, Appointment apps, AI ChatBot, AI based Voice agents, is considered Protected Health Information (PHI). Covered entities must obtain patient consent for the use of this information, and HIPAA requires that these authorizations, along with related documentation, be retained for at least 6 years from the date of creation or last effect.
Key Considerations for PHI Compliance:
- Website Initiated Contact Actions: If a patient enters health information, names, or contact info on your site, it is protected by HIPAA.
- 6-Year Rule: HIPAA regulations generally require maintaining documentation (including authorizations, policies, and procedures) for a minimum of 6 years.
- Retention Requirement: While the Privacy Rule doesn’t mandate specific retention times for medical records, it does for signed authorizations.
- State Law Exceptions: Some states have stricter retention laws that may require holding records for longer periods.
Type of typical consents:
- Consent for marketing communications.
- Consent for use of PHI to communicate with patients over non-encrypted channels (Excludes marketing communications)
- Consent for sending SMS/Text messages.
- Consent for sending Emails messages.
In addition to standard medical consent, healthcare providers often use these four distinct types of consent to manage digital communication and data usage. Under HIPAA, while some communication (like appointment reminders) may be permissible as “treatment,” most other digital interactions require explicit patient permission.
1. Consent for Marketing Communications
This is a written authorization required before using a patient’s Protected Health Information (PHI) for promotional purposes.
- What it covers: Communications that encourage a patient to purchase or use a product or service. Examples include promoting a third-party inhaler program or using patient testimonials/photos for advertising.
- Requirements: The form must be separate from treatment consents. It must clearly state if the provider is being paid by a third party to send the message.
2. Consent for Non-Encrypted Channels (Risk Acknowledgment)
HIPAA generally requires PHI to be encrypted during transmission. However, patients have the right to receive their health information via unencrypted channels (like standard personal email) if they choose.
- What it covers: Sending sensitive lab results, treatment plans, or billing details through standard email or SMS that lacks end-to-end encryption.
- Requirements: The provider must warn the patient of the security risks (e.g., that a third party could read the message). If the patient acknowledges these risks and still prefers the unencrypted method, the provider is not liable for breaches during transmission.
3. Consent for SMS/Text Messaging (TCPA & HIPAA)
Texting requires specific consent due to both HIPAA and the Telephone Consumer Protection Act (TCPA).
- What it covers: Any text sent to a patient’s mobile device, whether for reminders, satisfaction surveys, or health updates.
- Requirements: Patients must “opt-in” to receive texts. The consent should detail the frequency of messages, data rates, and clear instructions for opting out (e.g., “Text STOP to opt out”).
4. Consent for Email Communication
Similar to SMS, email consent ensures a patient wants to be contacted via this specific digital medium.
- What it covers: General health newsletters, practice updates, or portal notifications.
- Requirements: Consent should outline specifically which types of messages the patient will receive (e.g., billing vs. clinical updates). Documentation must be retained for at least six years to prove compliance during an audit.
Summary of Communication Consents
| Type | Purpose | Key Requirement |
|---|---|---|
| Marketing | Promotional outreach using PHI | Separate written authorization |
| Unencrypted | Discussion of clinical details | Mandatory “Duty to Warn” of security risks |
| SMS/Text | Mobile messaging | Explicit TCPA opt-in and STOP instructions |
| General digital updates | Specificity on message types and frequency |
